The hypothalamus comprises various nuclei and neuronal subpopulations that control fundamental homeostasis and behaviors. Even within a certain nucleus, different neuron types show particular gene expression patterns and spatial distribution characteristics, which are closely related to their functions. Our previous work revealed the novel neural stem cell, named hmRG in hypothalamus, that were similar to cortical oRG cells, plays an important role in hypothalamus development (Zhou et al., Nature Communications, 2020). However, spatiotemporal molecular characterization of hypothalamus development in humans is largely unexplored.
On Dec 7, 2021, a research paper leaded by Prof. Wu Qian at Beijing Normal University, in collaboration with Prof. WANG Xiaoqun at the Institute of Biophysics, Chinese Academy of Sciences, and Prof. Zhang Jun at Beijing Anzhen Hospital, Capital Medical University, entitled “Deciphering the spatial-temporal transcriptional landscape of human hypothalamus development” was published online in Cell Stem Cell. In this study, they applied single-cell transcriptional profiling to identify cell types in the developing human hypothalamus and revealed their developmental features.
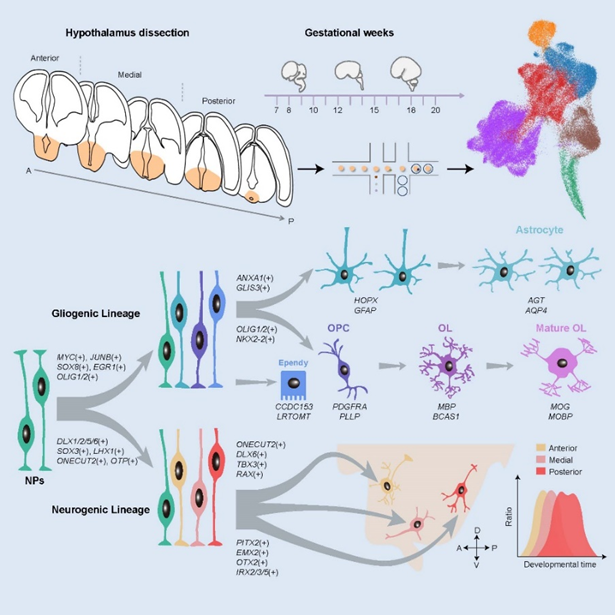
figure 1:Systematic characterization of the cell lineage and gene regulation of human hypothalamus development
Researchers revealed spatiotemporal transcriptome profiles and cell-type characteristics of human hypothalamus development and illustrated molecular diversity of neural progenitors and the cell fate decision, which is programmed by a combination of transcription factors. Different neuronal and glial fates are sequentially produced and showed spatial developmental asynchrony.
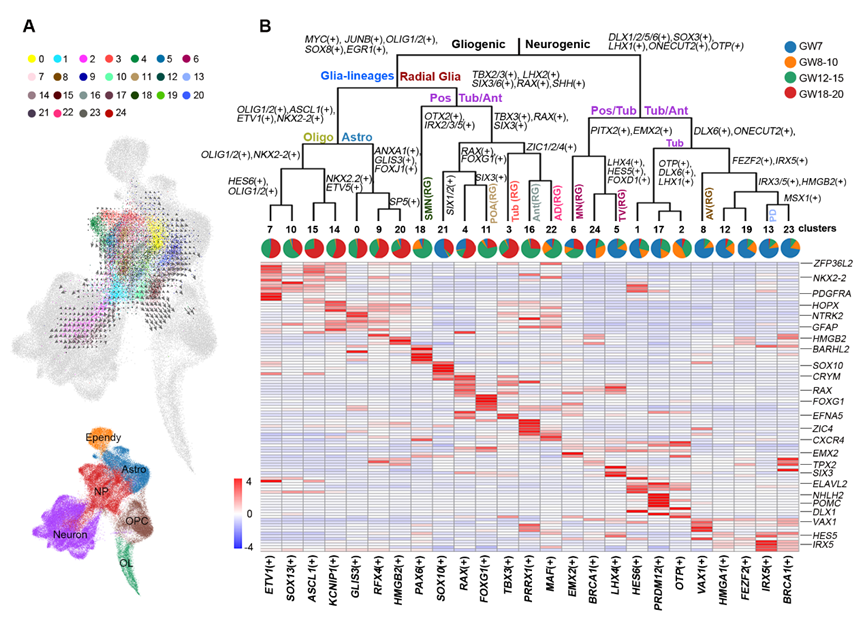
figure 2: Gene regulatory network of different neural stem cell subtypes
Furthermore, the researchers defined spatiotemporal transcriptional patterns of diverse neuronal subpopulations that would occupy specific nuclei during hypothalamic neurogenesis. In general, the timing of nucleogenesis in huma hypothalamus occurs in an anterior-to-posterior manner. Neurogenesis occurs at an early stage with different temporal waves and is gradually followed by gliogenesis proceeding with spatial asynchrony. They revealed heterogeneous oligodendrocyte (OL) lineage cells and traced their distinct developmental trajectory. Notably, early oligodendrocyte-neuronal interactions may have influenced neuronal differentiation by multiple signaling pathways, such as Wnt, Hippo and Integrin signals.
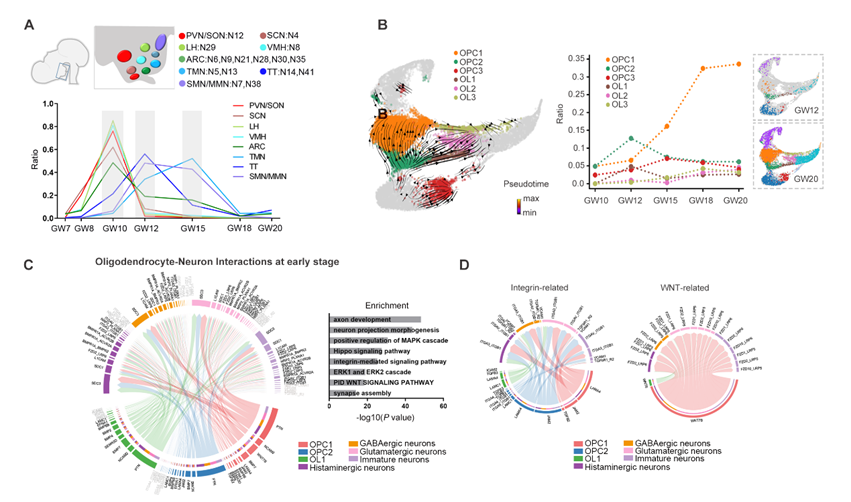
figure 3: The temporal characteristics of the development of hypothalamic neuron and glial cell subtypes
Finally, human hypothalamic gliogenesis occurs at an earlier stage of gestation and displays distinctive transcription profiles comparing with those in mouse. Compared with OLs in mice, they identified closely related human gene expression patterns that were particularly enriched in early oligodendrocyte lineage cells in human development.
Taken together, this study provides a comprehensive molecular landscape of human hypothalamus development at early- and mid-embryonic stages and a foundation for understanding its spatial and functional complexity.
Dr. Zhou Xin, Dr. Lu Yufeng, Zhao Fangqi are the first authors. This work was supported by National Basic Research Program of China, the Strategic Priority Research Program of the Chinese Academy of Sciences, the National Natural Science Foundation of China (NSFC) and Open Research Fund of the State Key Laboratory of Cognitive Neuroscience and Learning.